Ordinal Longitudinal Models (OLM)
- Extension of Wilcoxon test and Cox model to allow covariate adjustment + repeated measures
- Most flexible form uses a Markov process
- Demonstrated to handle within-pt serial correlation almost perfectly in multiple RCTs
- Better modeling of intra-pt correlation
- Elegantly handles missing components + absorbing states precluding pt scale assessment
- Death and need for rescue therapy accounted for
OLM, continued
- Huge variety of clinical readouts
- Pr(transitioning to state
- Pr(being at severity y or worse as a function of time, tx)
- Mean time in any set of states
- Treatment difference in expected time in specified states (like
- Pr(transitioning to state
- Generalizes Wilcoxon test, Cox model, recurrent event analysis, and longitudinal analysis
Popular OLM Readout
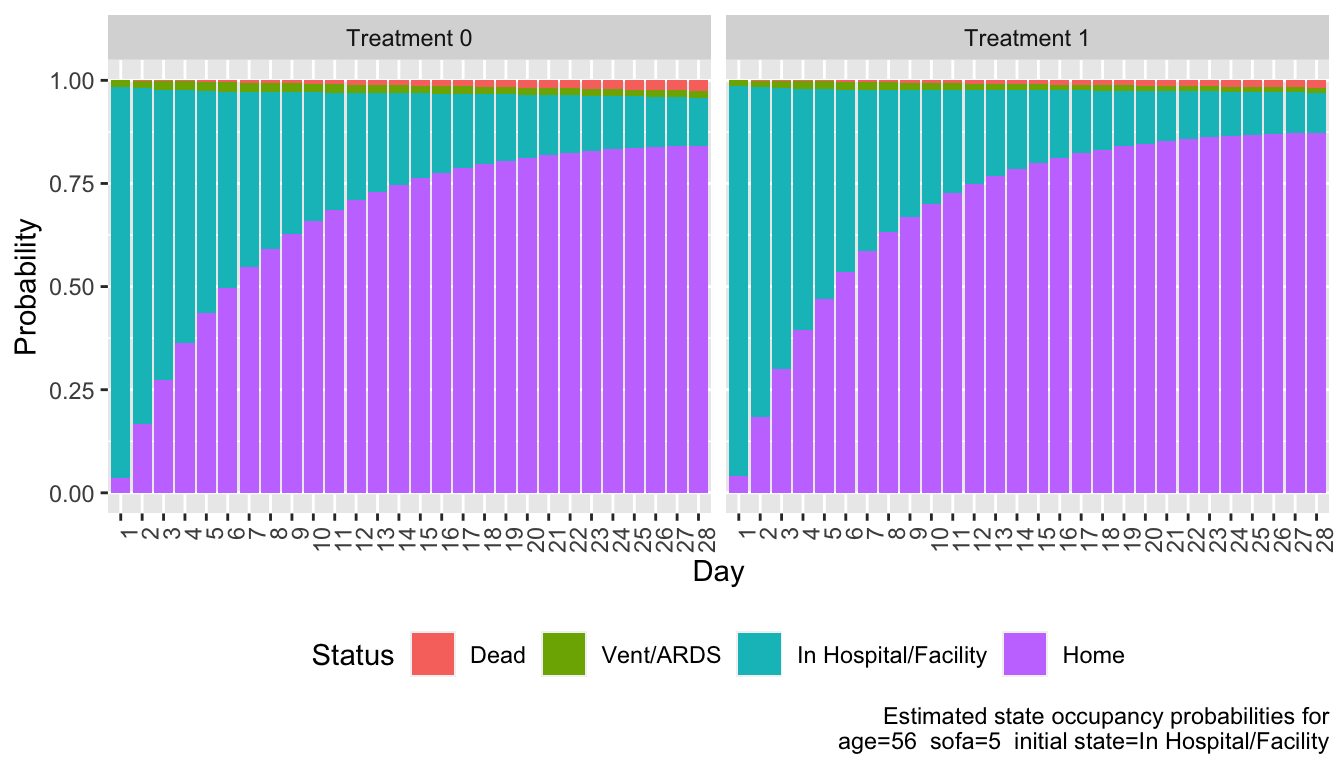
OLM, continued
- OLM works for tx that improves pt condition as well as for tx for slowing progression
- Detailed case study with complete R code at hbiostat.org/rmsc/markov
- OB project underway to reanalyze an ALS trial using OLM
Time Savings
- Dickson, Wessels, Dowsett, Mallinckrodt, Sparks, Chatterjee, Hendrix J Prev Alzheimer's 2023
- Analyses single composite outcome measure or stat summaries of separate measures
- For degenerative disease, not for treatments that improve pts over their baseline state
- Assumes follow-up time is sufficiently long that almost all control group pts fair poorly
TS, continued
- TS is essentially
- Based on linear interpolation on estimated means
- TS cannot account for death or need for rescue therapy
Comparisons of TS and OLM
- OLM assumes consensus in severity ordering of outcome states for a single assessment time
- Uses only the worst condition suffered by the patient on a given day
- Expect OLM to have greater power than TS due to
- More use of all the raw data over the entire time course
- OLM accounts for how close to failure were active arm pts before the last follow-up
TS vs. OLM, continued
- OLM allows for absorbing states/terminating events that preclude patient scale assessment
- OLM explicitly accounts for deaths in an interpretable fashion
- Contrast that with counterfactuals and competing risk analysis
- TS cannot easily borrow information
- OLM has been implemented in both frequentist and Bayesian models
- OLM can formally assess how tx affects different outcome components
Composite Outcome Scales
- Choice of scales is very important
- Gold standard is pt utility for current status
- OLM approximates the gold standard
- Several ways to combine multiple scales
- TS approach using global statistical summary
- is not clinically interpretable at a given time
- cannot handle deaths
- difficult to handle missing component data