What Are the Most Common Causes of Equivocal Results?
- Over-optimistic power calculation
- using an effect size > clinically relevant
- if effect is "only" clinically relevant, likely to miss it
- Fixed sample size
- Losing power in the belief that α is something that should be controlled when taking multiple data looks
- Insensitive outcome measure
Another Common Outcome
- Little evidence the treatment works
- Usually the study could have been stopped much sooner
- Q: How far along can a trial be and still have a good chance for a positive study if difference is negative?
- A:
The Problem With Sample Size
- Frequentist (traditional statistics): need a final sample size to know when α is "spent"
- Fixed budgeting also requires a maximum sample size
- Sample size calculations are voodoo
- arbitrary α, β, effect to detect Δ
- requires accurate σ or event incidence
- Physics approach: experiment until you have the answer
Is a Sample Size Calculation Needed?
- No, if using a Bayesian sequential design
- With Bayes, study extension is trivial and requires no α penalty
- logical to recruit more patients if result is promising but not definitive
(0.7 < P(benefit) < 0.95) - analysis of cumulative data after new data added merely supersedes analysis of previous data
- logical to recruit more patients if result is promising but not definitive
Is α a Good Thing to Control?
- α = type I assertion probability = P(p < 0.05 | H0) typically
- It is not the probability of making an error in acting as if a treatment works
- It is the probability of making an assertion of efficacy (rejecting H0) when any assertion of efficacy would by definition be wrong (i.e., under H0)
- α ⇑ when as # data looks ⇑
Bayesian vs. Frequentist Designs
- Controlling α leads to conservatism when there are multiple data looks
- Bayesian sequential designs: expected sample size at time of stopping study for efficacy/harm/inefficacy ⇓ as # looks ⇑
- Example
- Frequentist group-sequential design vs. Bayesian continuous sequential design
- Decision rules for non-trivial efficacy, inefficacy, similarity
- Average stopping time for frequentist: 273 treated patients
- Bayesian: 113
What Should We Control?
- The probability of being wrong in acting as if a treatment works
- This is one minus the Bayesian posterior probability of efficacy (probability of inefficacy or harm)
- Controlled by the prior distribution (+ data, statistical model, outcome measure, sample size)
- Example: P(HR < 1 | current data, prior) = 0.96
⇒ P(HR ≥ 1) = 0.04 (inefficacy or harm)
The Shocking Truth of Bayes
- It controls the reliability of evidence at the decision point
- Not the pre-study tendency for data extremes under an unknowable assumption
- Simulation examples: bit.ly/bayesOp
Avoid Low-Resolution Outcome Variables
- When an event has multiple severity levels or is recurring:
- Significant lost of power with Y = time to first event
- Need 462 events to estimate a hazard ratio to within a factor of 1.20 (from 0.95 CI)
- Need 384 patients to estimate a difference in means to within 0.2 SD (n = 96 for Xover design)
General Outcome Attributes
- Timing and severity of outcomes
- Handle
- terminal events (death)
- non-terminal severe events (stroke)
- recurrent events (seizures)
- Break the ties; the more levels of Y the better
- Maximum power when no two patients have the same Y
What is a Fundamental Outcome Assessment?
- In a given week or day what is the severity of the worst thing that happened to the patient?
- Expert clinician consensus of outcome ranks
- Spacing of outcome categories irrelevant
- Can code common co-occurring events as worse event
Clinical Readouts
- Can translate an ordinal longitudinal model to obtain a variety of estimates
- time until a condition
- expected time in state
- probability of something bad or worse happening to the pt over time, by treatment
- Bayesian partial proportional odds model can compute the probability that the treatment affects mortality differently than it affects nonfatal outcomes
Examples of Longitudinal Ordinal Outcomes
- 0=alive 1=dead (absorbing state; cease data collection)
- censored at 3w: 000
- death at 2w: 01
- longitudinal binary logistic model OR ≅ HR
Ordinal Example
- 0=OK, 1=seizure, 2=status epilepticus, 3=hospitalized, 4=treatment failure, 5=death
- Treatment failure: escape criterion: e.g.
- Absorbing states: treatment failure, death
- Seizure at 3d, status at 5d, hospitalized at 9d-11d, death at 12d: 001020003335
Statistical Model
- Proportional odds ordinal logistic model with covariate adjustment
- Handles intra-patient correlation with a Markov process + random intercepts
- Extension of binary logistic model
- Generalization of Wilcoxon-Mann-Whitney Two-Sample Test
- No assumption about Y distribution for a given patient type
- Does not use the numeric Y codes
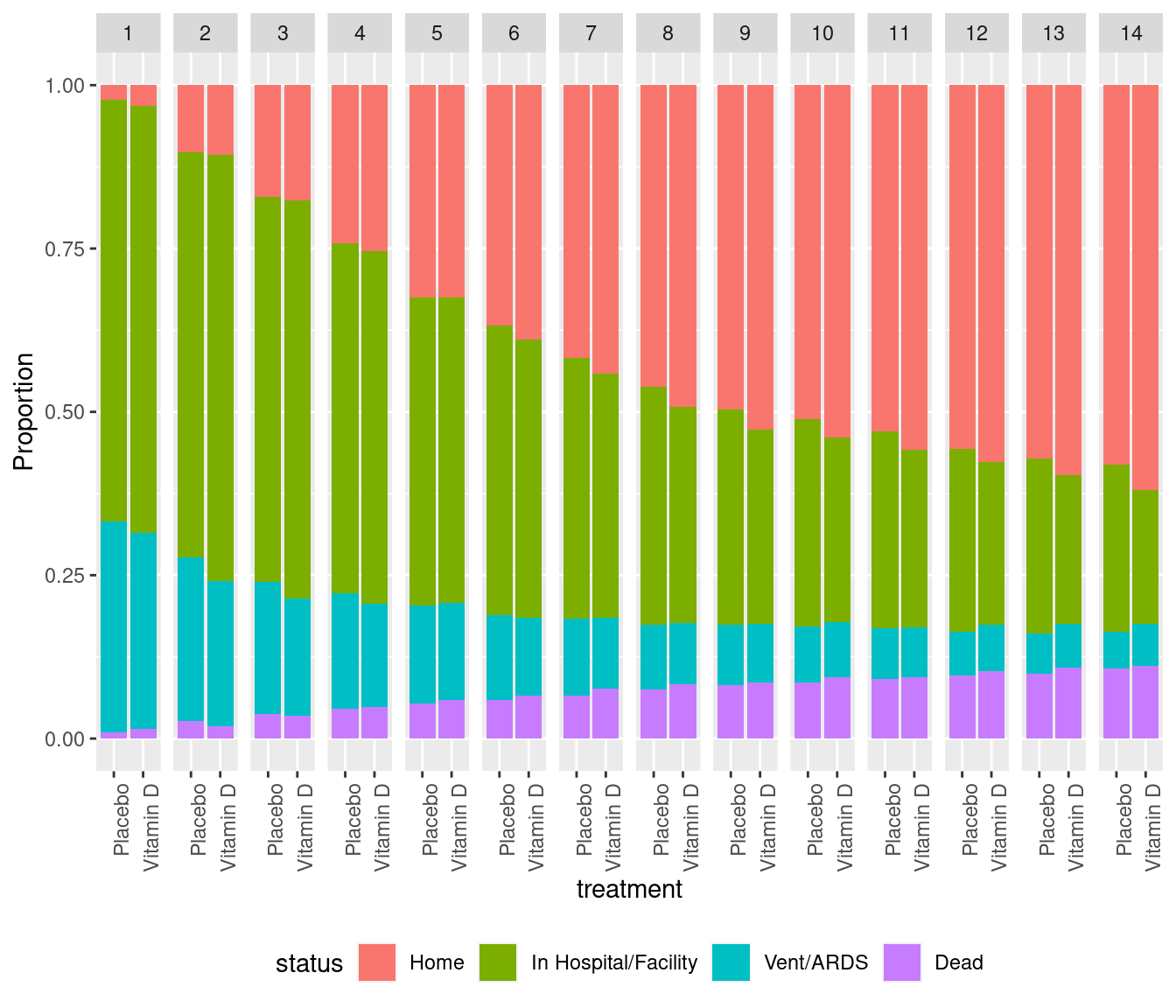
Example Study
- VIOLET (Petal Network, NEJM 381:2529, 2019)
- Early high-dose vitamin D
- Primary endpoint: mortality (slight evidence for increase with D
- Ordinal endpoint collected each day for 28 consecutive days
VIOLET Day 1 - 14 Ordinal Outcomes
Statistical Power from Using More Raw Data
- Simulation of VIOLET-like studies
| Method | Power |
|---|---|
| Time-to-recovery analysis with Cox model | 0.79 |
| Wilcoxon test of vent/ARDS-free days with death=-1 | 0.32 |
| Longitudinal ordinal model | 0.94 |
Details at hbiostat.org/R/Hmisc/markov/sim.html
General Principles for Choosing Outcome Measures
- Increase power by breaking ties &
- Get close to the raw data
- Relevant to patients
- Respect timing and severity of outcomes
- Can do automatic risk/benefit trade-offs by including safety events in an ordinal outcome scale
Take Home Messages
- Don't take sample sizes seriously; consider sequential designs with unlimited data looks and study extension
- α is not a relevant quantity to "control" or "spend" (unrelated to decision errors)
- Choose high-resolution high-information Y
- Longitudinal ordinal Y is a general and flexible way to capture severity and timing of outcomes
- Always adjust for strong baseline prognostic factors
Quickest Path to Generation of Reliable Knowledge
- When there are two treatment arms
- Use RWD to
- refine the within-patient correlation pattern to model
- estimate variability for (Bayesian) power calculations
- 1:1 randomization with only indirect use of RWD
- Fully sequential trial with multiple decision rules that stop when evidence is strong
- High-resolution longitudinal Y
More Information
hbiostat.org/endpointdatamethods.orghbiostat.org/bbr/alphahbiostat.org/bayes/designfharrell.com/post/nfl